Scientists Use RNA 'Barcodes' to Map Thousands of Brain Connections at Single-Synapse Precision
University of Illinois researchers published the first technology to simultaneously map thousands of neural connections, revealing previously unknown wiring in the mouse brain and opening a new path to understanding Alzheimer's.
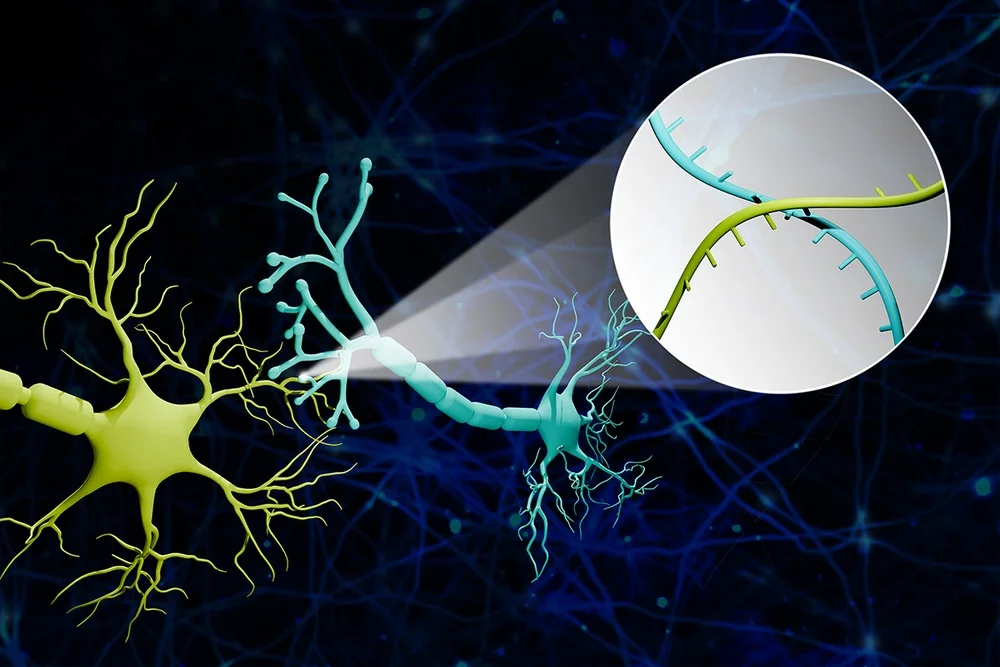
Scientists at the University of Illinois at Urbana-Champaign have developed a transformative technology called Connectome-seq that uses unique RNA "barcodes" to map thousands of neural connections in the brain simultaneously at single-synapse precision — a capability that previously did not exist in any technology available to neuroscientists. The work, published in Nature Methods on March 13, 2026, and highlighted in ScienceDaily this week, was led by Professor Boxuan Zhao from the Department of Cell and Developmental Biology. In initial experiments using mouse brains, Connectome-seq revealed previously unknown connectivity patterns between distinct types of brain cells, including connections that had not appeared in any prior anatomical atlas.
The challenge of mapping how neurons are connected — the problem known as connectomics — has long been one of the most technically demanding frontiers in biology. A human brain contains roughly 86 billion neurons connected by an estimated 100 trillion synapses. Previous mapping approaches, including electron microscopy, could produce exquisitely detailed images of individual synapses but were agonizingly slow: mapping even a tiny cubic millimeter of brain tissue could take years. Connectome-seq bypasses this bottleneck entirely by converting the problem of neural mapping into a high-speed sequencing task. "We translated the neural connectivity problem into a sequencing problem," Zhao said. "Our technology enables simultaneous mapping of thousands of neural connections with single-synapse resolution — a capability that doesn't exist in any current technology."
The mechanism works by assigning each neuron in a sample a unique RNA barcode — a short, distinctive genetic tag. Specialized proteins carry these barcodes from the neuron's main body outward to its synaptic terminals, the tiny structures where one neuron physically touches and communicates with another. Researchers then isolate those synapses and use high-throughput sequencing to read which barcode pairs are found together at each contact point. Because barcodes from connected neurons accumulate at the same synapses, the sequencing data reveals which neurons are wired to which — across thousands of connections at once. The researchers validated the approach in the mouse pontocerebellar circuit, successfully mapping over 1,000 neurons and identifying connection patterns that had not previously been documented.
The discovery of unexpected connectivity in the adult mouse brain hints at how much of the brain's fundamental wiring remains unknown despite decades of research. Zhao's team found neuron types connecting to partners that anatomists had not previously associated, suggesting that the brain's architecture may be substantially more complex and variable than existing models predict. The implications extend beyond basic neuroscience. Brain disorders including Alzheimer's disease, Parkinson's disease, schizophrenia, and autism spectrum disorders are all believed to involve disruptions in the way neurons are connected. A technology capable of mapping those connections at scale could accelerate the identification of which circuits go wrong in each condition — and potentially identify the disruptions before symptoms appear.
The publication has generated considerable interest across neuroscience and genomics communities for its combination of conceptual simplicity and technical power. Unlike approaches that require radioactive labeling, viral injection, or electron microscope preparation — all of which are slow, expensive, and difficult to scale — Connectome-seq uses standard molecular biology reagents in a workflow compatible with existing sequencing platforms. Zhao and colleagues are already working to adapt the technology for human brain tissue obtained from biobanks, with the goal of eventually comparing connectivity maps from healthy brains with those from donors who had Alzheimer's or other neurological disorders. If successful, it could provide the first high-resolution picture of how the human brain's wiring degrades in disease.
Originally reported by University of Illinois / Nature Methods.