Scientists Solve 100-Year Mystery of How Cinchona Trees Make Quinine, Opening Path to Synthetic Production
Researchers at the Max Planck Institute identified the complete enzyme pathway that produces quinine in tree bark, including a novel intermediate compound called cinchonium never seen before in nature.
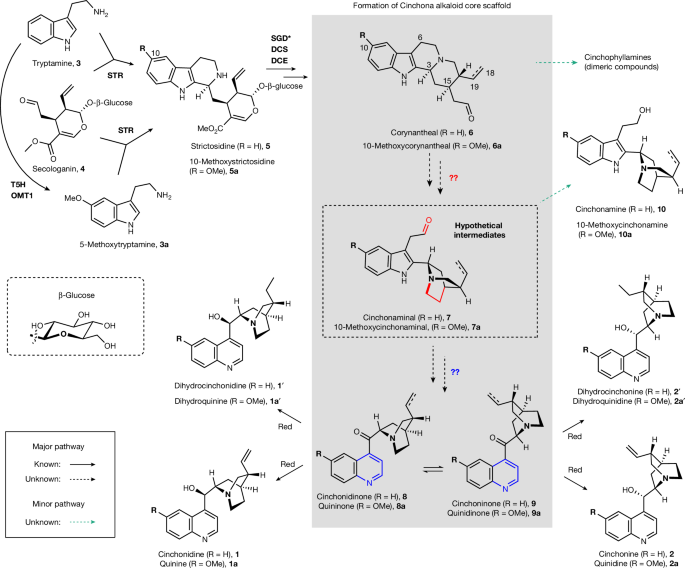
Scientists have solved a 100-year-old botanical mystery by identifying the complete set of enzymes that trees use to synthesize quinine, the antimalarial compound and flavor ingredient in tonic water that has been derived from the bark of South American cinchona trees since the 17th century. The findings, published in Nature by researchers led by Sarah E. O'Connor at the Max Planck Institute for Chemical Ecology in Germany and C. Robin Buell at the University of Georgia, reveal an unexpectedly complex and previously unknown enzymatic pathway that plants evolved over millions of years to produce the distinctive quinoline-quinuclidine scaffold at the heart of quinine and its closely related cousins — collectively called cinchona alkaloids.
The medical and commercial importance of quinine and its derivatives is difficult to overstate. Quinine was the only effective treatment for malaria for more than three centuries before synthetic alternatives were developed in the 20th century. Chloroquine and hydroxychloroquine, the synthetic antimalarials that largely displaced it and later attracted attention as potential COVID-19 treatments, are structural analogs derived from quinine's chemistry. The cinchona alkaloids also include quinidine, still used to treat certain cardiac arrhythmias, and quinine itself remains a second-line treatment for drug-resistant malaria in regions where newer drugs have failed. Despite this medical importance, the full biosynthetic pathway by which cinchona trees make these compounds had never been characterized — a gap that prevented chemists from engineering microbes or plants to produce them more efficiently.
The research team discovered several previously unknown enzymes, including a transferase, an oxoglutarate-dependent dioxygenase, and a cytochrome P450, that work in sequence to build the alkaloids' distinctive bicyclic ring structure. Most surprising was the identification of an intermediate compound the researchers named cinchonium — a quaternary amine produced by an unusual enzymatic cyclization step that had no precedent in known plant biochemistry. "One of the most exciting moments was finding this completely novel intermediate," O'Connor said. "It tells us that plant chemistry continues to surprise us even in species we've studied for centuries."
The practical payoff is the potential to produce quinine and related alkaloids through synthetic biology rather than by harvesting and processing cinchona bark — a process currently centered in countries including Peru, the Democratic Republic of Congo, and Indonesia, where the trees are grown on plantations. Bark extraction yields are low and the alkaloids are present in minute quantities, making production expensive and geographically constrained. With the complete enzymatic pathway now mapped, researchers can in principle engineer bacteria or yeast to carry out the same biosynthetic steps in fermentation tanks, producing unlimited quantities without deforestation or geographic dependency. O'Connor's group has already demonstrated proof-of-concept in E. coli and in the model plant Nicotiana benthamiana, though yields need substantial improvement before industrial-scale production becomes feasible.
The discovery also has implications for drug discovery more broadly. Cinchona alkaloids are structurally unusual in ways that pharmaceutical chemists have long wanted to exploit but have been unable to access through conventional synthesis. Now that the biosynthetic toolkit is fully characterized, researchers can use the enzymes to produce novel analogs of quinine with modified structures — opening an enormous unexplored chemical space of potential antimalarial agents, antiarrhythmic drugs, and other bioactive molecules. The Max Planck team has already begun a collaboration with the Medicines for Malaria Venture, a nonprofit drug development organization, to screen novel cinchona alkaloid analogs against drug-resistant Plasmodium falciparum strains that have become endemic in parts of sub-Saharan Africa.
Originally reported by Nature.