Scientists Discover 'Bone Switch' GPR133 That Rebuilds Weakened Bones, Offering Hope for Osteoporosis
Leipzig University researchers identified a molecular receptor that controls bone formation and showed a new compound called AP503 can dramatically boost bone density in mice, pointing toward a new class of osteoporosis treatment.
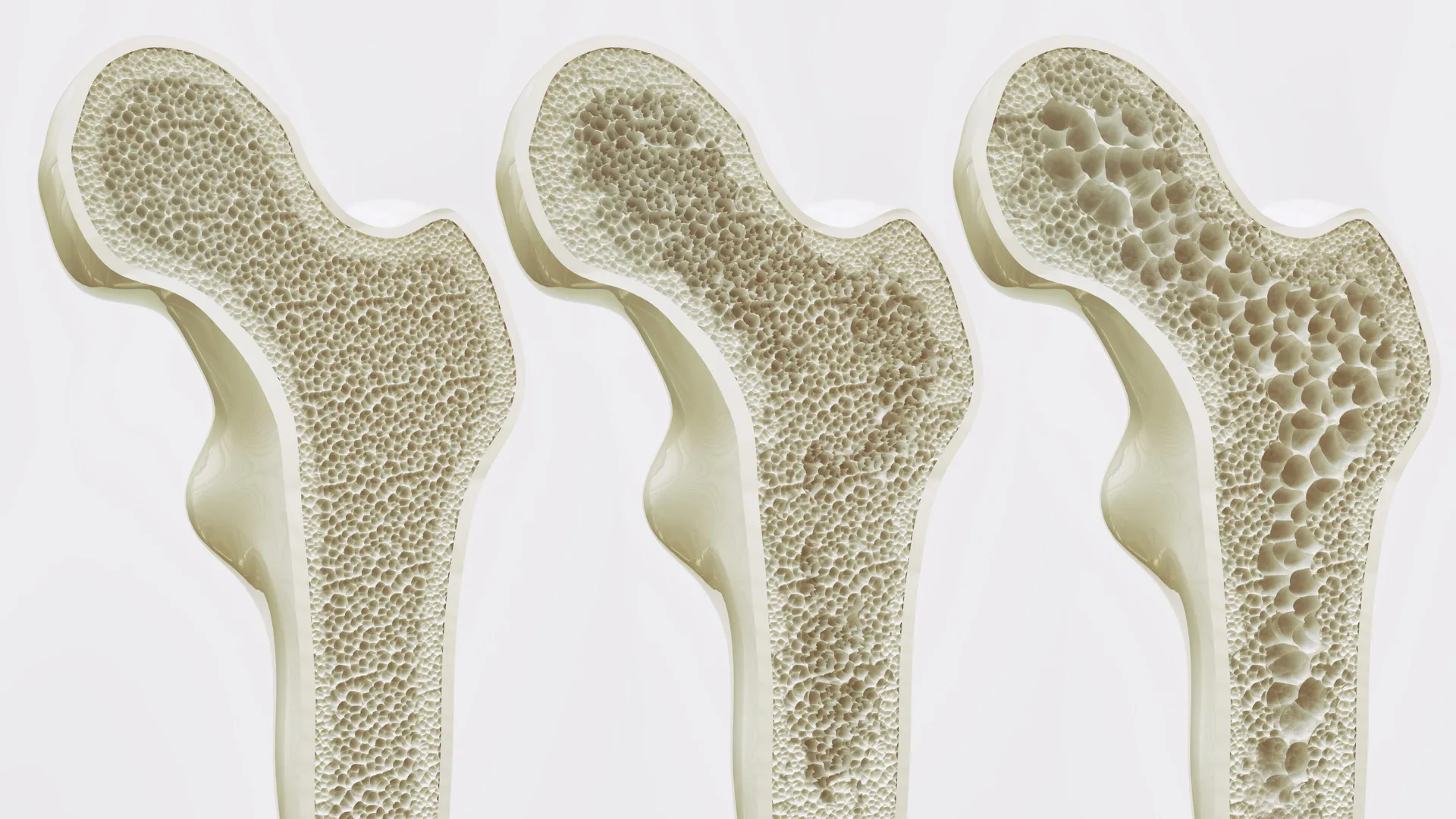
Researchers at Leipzig University in Germany have identified a molecular receptor called GPR133 that functions as a master switch controlling bone formation, and have shown that a new compound called AP503 can activate this switch to dramatically increase bone density in mice — findings that point toward an entirely new class of treatment for osteoporosis, a condition that affects more than 200 million people worldwide and causes millions of fractures each year.
Osteoporosis occurs when the body breaks down bone faster than it can rebuild it, leaving the skeleton increasingly fragile and vulnerable to fractures from minor falls or everyday activities. Existing treatments either slow bone loss by inhibiting the cells that break bone down, or stimulate bone formation through hormonal mechanisms. The GPR133 discovery opens a third avenue: directly activating a cellular receptor that appears to be a fundamental regulator of bone-building activity.
The Leipzig team found that GPR133, which belongs to a family of receptors called adhesion G protein-coupled receptors, is expressed on osteoblasts — the cells responsible for laying down new bone tissue. When GPR133 is activated, osteoblasts increase their bone-forming activity significantly. The researchers tested AP503, a synthetic compound designed to bind to and activate GPR133, in mouse models of osteoporosis and observed substantial improvements in bone density compared to untreated animals.
The significance of the discovery lies partly in identifying a specific molecular target that can be addressed with precision. Many existing osteoporosis treatments have side effects that limit their long-term use — some bone-formation-stimulating drugs, for example, can only be used for limited periods before they begin to have adverse effects on the heart or other systems. A GPR133-targeted therapy might offer a more specific mechanism with fewer off-target effects, though that would need to be demonstrated in subsequent human trials.
The researchers emphasized that the findings are at an early stage and significant work remains before a GPR133-targeted therapy could be available for patients. The next steps include testing whether the effects seen in mice translate to larger animals and eventually humans, and whether AP503 or similar compounds are safe for human use. Drug development from this point typically takes ten or more years and involves extensive clinical trials. Nevertheless, the identification of GPR133 as a bone formation switch gives pharmaceutical researchers a clearly defined molecular target to pursue with new drug development efforts.
Originally reported by ScienceDaily.