Scientists Unlock Secret Behind Nature's 'Proton Highway' in Major Physics Breakthrough
Researchers freeze key molecular structures to reveal how electrical charges move so efficiently through biological systems and advanced technologies.
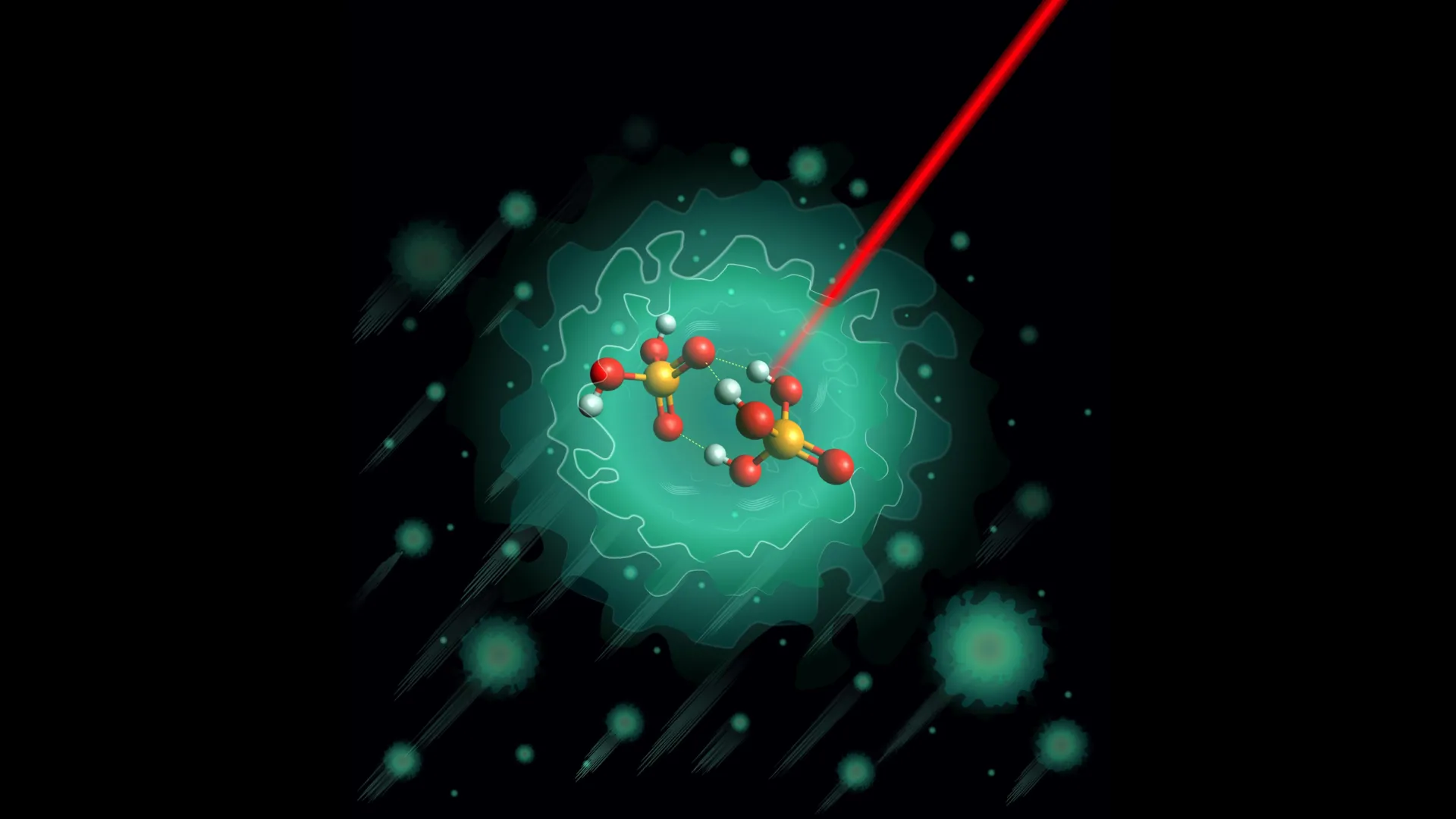
Scientists have made a groundbreaking discovery about how nature efficiently transports electrical charges through biological systems, uncovering the molecular secrets behind what researchers call the "proton highway." By freezing key molecular pairs to extremely low temperatures, physicists have revealed that phosphoric acid forms a single, highly stable structure that contradicts previous theoretical predictions and explains the remarkable efficiency of charge transport in living organisms.
The research, focusing on phosphoric acid and related compounds found throughout biological systems, addresses a fundamental question about how protons move so quickly through materials like DNA, RNA, and cell membranes. These compounds are also central to energy storage in ATP molecules and are widely used in technological applications such as batteries and fuel cells because of their exceptional proton-conducting properties.
Using advanced experimental techniques, researchers from the Department of Molecular Physics at the Fritz Haber Institute created the deprotonated dimer H3PO4·H2PO4- in laboratory conditions and cooled it to just 0.37 degrees above absolute zero inside helium nanodroplets. At these extreme temperatures, molecular vibrations are nearly eliminated, allowing scientists to analyze the structure with unprecedented precision using infrared spectroscopy combined with quantum chemical calculations.
The experimental results revealed an unexpected finding that challenges existing theoretical models. While computer simulations had predicted two equally likely molecular configurations, the actual measurements showed only one stable structure featuring three hydrogen bonds connected through a shared oxygen atom. This rigid arrangement creates specific pathways for proton movement that appear to be universal across similar molecular systems.
The discovery has significant implications for understanding fundamental biological processes and developing better energy technologies. The specific hydrogen-bond network identified in this research may serve as a template for designing more efficient proton-conducting materials for next-generation batteries and fuel cells. Additionally, the findings provide new insights into how cellular energy production and genetic information storage systems achieve their remarkable efficiency through optimized molecular structures.
Originally reported by ScienceDaily Physics.